
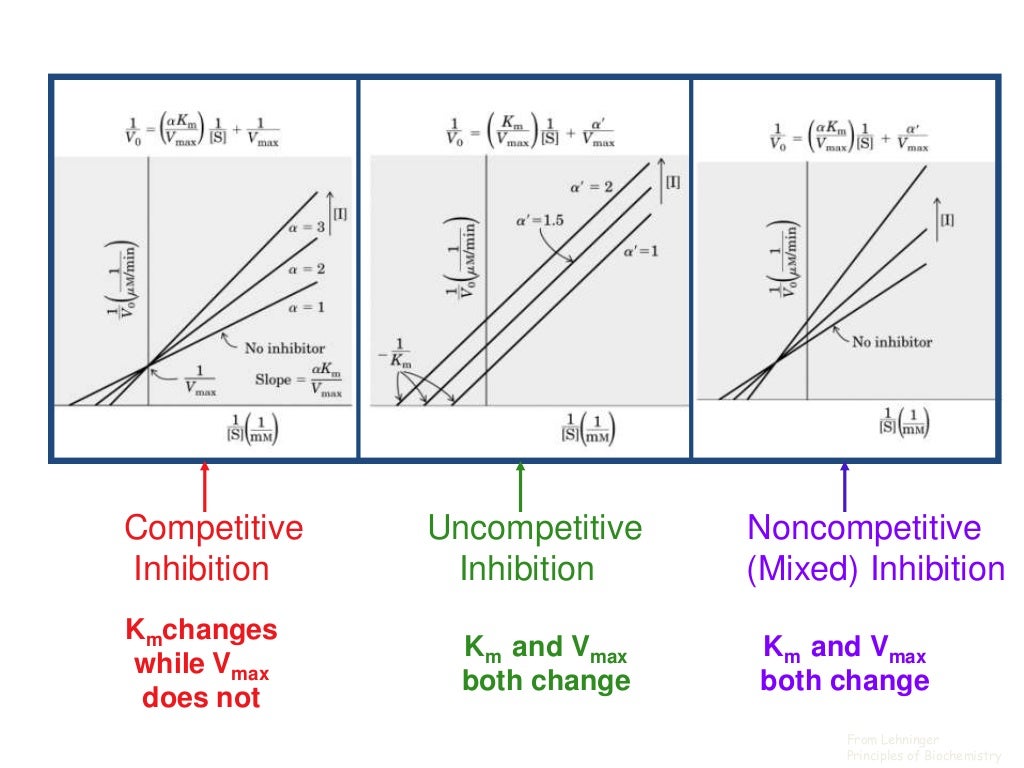

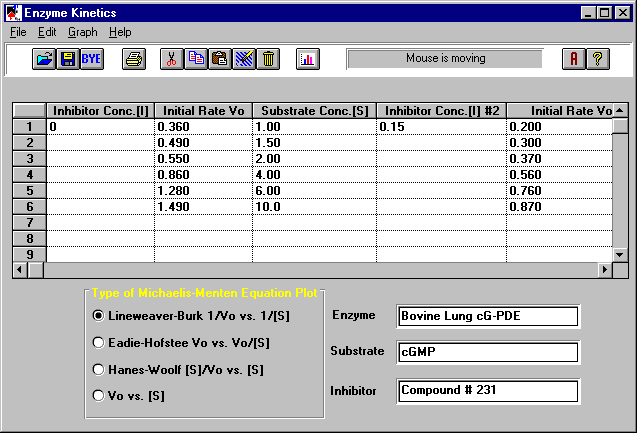
The kinetics of biological processes appear to differ only quantitatively from those of in vitro enzyme-catalyzed reactions. The reactive configuration of the enzyme is apparently possessed in only a relatively narrow temperature band, being lost at both high and low temperatures. Upon the basis of this interpretation, values of the entropy changes that are calculated theoretically are found to be in agreement with those calculated from the experimental data. It is suggested that at low temperatures the formation of intramolecular hydrogen bridges converts reactive enzyme particles to a catalytically inactive condition, in which the active centers either lose their specific configuration or are no longer exposed to the substrate. The results of applying these relations to biochemical processes support the contention that in the lower temperature range of enzyme activity a thermodynamic equilibrium exists between catalytically active and inactive forms of the enzyme. Used in conjunction with the formulation of the theory of absolute reaction rates this empirical relation facilitates the determination of the thermodynamic functions. It is found empirically that a simple modification of the usual theoretical kinetic formulation (in which a transformation in the temperature scale is made) describes the temperature dependence of a wide variety of biochemical processes with a greater accuracy than hitherto achieved.

 RSS Feed
RSS Feed
